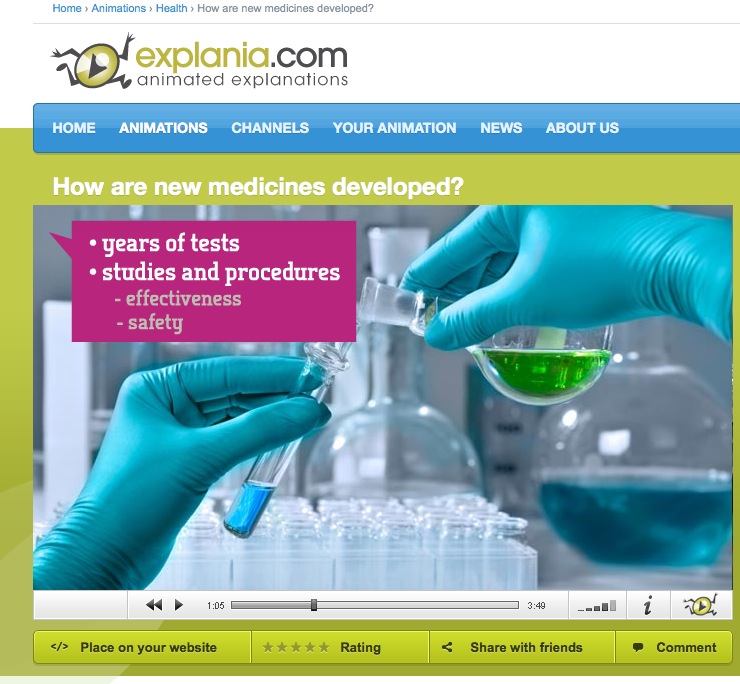
Video: How are new medicines developed?
This short film explains how innovative medicines are developed and brought to market, and how they turn into cheaper generic medicines after some years. Researchers can get to the registration phase only 1 or 2 substances out of 10,000 preselected molecules. Then it takes 12-13 years of tests and studies to prove its effectiveness and safety, first in test tubes, then on healthy volunteers and finally on thousands of patients. This is a risky investment for a pharmaceutical company. Aften the temporary patent expires, other companies can copy the active ingredient and sell it without having to do any research and testing. Students can listen to advanced vocabulary about business management and pharmacy.
-
Idioma:
- Inglés
Recurso educativo
-
Aún no hay comentarios, ¡comparte tu opinión! Inicia sesión o Únete a Tiching para poder comentar







¿Dónde quieres compartirlo?
¿Quieres copiar el enlace?